Project 1: Cortical circuits for sensory predictive coding and mismatch negativity. Sensory processing in the cerebral cortex is significantly modulated by context. Interactions among top-down, bottom-up, and local neuronal connections serve to modulate the processing of sensory inputs based on experienced context. This modulation involves the reduction of responses to predictable stimuli (e.g. repetition suppression) and enhancement of neuronal responses to unexpected stimuli (e.g. “prediction errors”), which then update circuit connections to dynamically shape how an individual perceives and acts on the sensory world. Though basic, this rapid, dynamic modulation is critical for survival. Further, people with major psychotic diseases like schizophrenia exhibit aberrant “prediction error” responses to unexpected stimuli, as indexed by the EEG – based “mismatch negativity” biomarker. Currently, the essential cortical circuitry underlying the generation of sensory prediction error is unknown.

The Hamm lab harnesses circuit neuroscience tools such as optogenetics, two-photon calcium imaging, and extracellular electrophysiology in awake mice to provide a comprehensive understanding of how cortical circuits, both local and interregional, encode context to modulate processing of incident stimuli. Subprojects address how glutamatergic inputs from the prefrontal cortex vs acetylcholinergic inputs from the basal forebrain modulate sensory cortices at various circuit levels. Special focus will be put on local interneuron subpopulations and interlaminar dynamics within visual and auditory cortices, as well as neuro-oscillatory signatures of prediction errors which at once deepen our understanding of circuit dynamics as well as serve as translational hooks for relating findings in rodents to human EEG measurements in a clinical setting. Results from this project will provide novel insights for future precision treatments of schizophrenia.
Publications for project 1:
Van Derveer, A., Ross, J.M., Hamm. J.P. (2023) Robust multisensory deviance detection in the mouse parietal associative area. Current Biology-accepted, in press. see BioRxiv
Gallimore, C.G., Ricci, D., Hamm. J.P. (2023) Spatiotemporal dynamics across a neocortical column support a predictive coding framework for interpreting visual mismatch responses. Cerebral Cortex. 33(15): 9417–9428. DOI: 10.1093/cercor/bhad215
Hamm, J.P., Shymkiv, Y., Han, S., Yang, W. Yuste, R. Cortical ensembles selective for context. (2021). Proceedings of the National Academy of Sciences of the United States of America (PNAS). 118(14):e2026179118. DOI: 10.1073/pnas.2026179118.
Ross, J.M., Hamm, J.P. (2020) Cortical Microcircuit Mechanisms of Mismatch Negativity and Its Underlying Subcomponents. Front Neural Circuits. 14:13. DOI: 10.3389/fncir.2020.00013
Funding for project 1: “Circuits for deviance detection in V1”, National Institutes of Health. R01EY033950. ($250,000 per year direct). 2022-2027
“Fronto-sensory circuit mechanisms of perceptual novelty processing“, NIMH R00-MH115082, PI: J.P. Hamm ($250,000/year total). 2017-2022
“Mapping the role of basal forebrain projections to visual cortex in novelty processing”, Whitehall Foundation, PI: J.P. Hamm ($75,000/year direct). 2020-2022
“Transcriptomic Profiling of a Novel Subtype of Cortical Neurons Selective for Sensory Prediction Errors”, Brain and Behavior Research Foundation (BBRF) Young Investigator Grant (NARSAD; 2 years; $35,000 per year, direct). 2021-2023
Project 2: The role of microglia-neuron interactions in sculpting cortical circuits during adolescence
A basic understanding of neuron-glia interactions is key to linking altered immune function to disrupted neural circuitry and cognition present in major psychiatric diseases. Microglia are a resident immune cell in the cerebral cortex, and one of their main functions under physiological conditions is to modify synaptic connections among neurons. How these activities extend to influence higher-order functional networks in cortical circuits is not clear, particularly in brain regions crucial for cognitive function, such as the prefrontal cortex (PFC).
A key may lie in how neural circuit synchrony stimulates nearby microglial cell motility – i.e. active extension and retraction of fine cellular processes – and, specifically, in how this relationship changes throughout adolescence, a critical period for the development of PFC and higher cognition. Further, sex differences have been established in some aspects of microglial function. Clarifying how sex modulates the role of microglia in PFC circuit development is essential, especially given the dramatic sex differences in vulnerability to adolescent onset of psychiatric diseases such as schizophrenia.
The goal of the current project is to obtain a basic understanding of glial-neuronal-circuit interactions in the mammalian prefrontal cortex. The planned approach (Aim 1) employs three-dimensional two-photon microscopy and single neuron optogenetics in awake mouse medial prefrontal cortex (mPFC) to elucidate how the structural dynamics of microglial cells are driven by neuronal activity and oscillatory synchrony in local circuits. This will be examined at distinct time windows from pre-adolescence into early adulthood and compared between males and females. (Aim 2,3) To test whether, how, and when microglia activity is necessary for the establishment of adult mPFC function, microglia will be selectively eliminated during restricted windows during adolescence and early adulthood using a pharmacological strategy. Then in adulthood, sex- and adolescent-period specific effects on the development of i) (Aim 2) spatiotemporal circuit dynamics in mPFC (functional network clustering, gamma oscillations, theta-gamma coupling) will be measured using two-photon calcium imaging and dense electrical recordings and ii) (Aim 3) PFC-dependent cognition will be assessed with an established odor-based attentional set-shifting task.
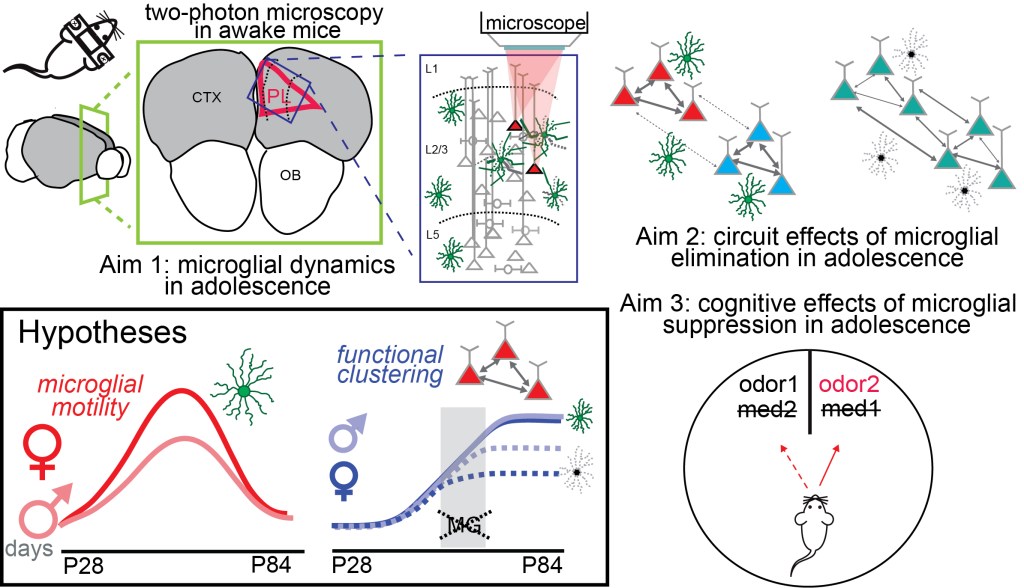
Funding for project 2: “Sex differences in microglia-neuron-circuit interactions in adolescence” NIMH R01-MH128176-01, PI: J.P. Hamm ($250,000/year direct). 2022-2027
